Experts are now planning to mix Covaxin and Covishield doses for ‘better efficacy’; Green light for trials
The SEC has recommended that permission be granted to Christian Medical College (CMC), Vellore, for conducting a clinical trial on mixing of Covaxin and Covishield.
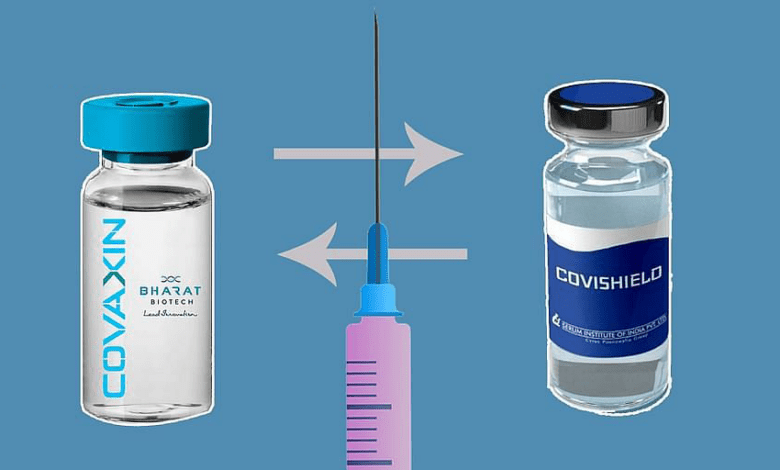
A Subject Expert Committee of the Central Drugs Standard Control Organisation (CDSCO) on Thursday recommended conducting a study on mixing doses of Covishield and Covaxin vaccines.
Apart from this, the expert panel also recommended mixing of Bharat Biotech’s Covaxin and a nasal vaccine candidate, while saying they are looking at trials for Biological E’s Covid-19 vaccine on children.
However, a final approval from the Drugs Controller General of India (DCGI) is awaited.
As per sources, the application of the interchangeability study protocol of Covishield and Covaxin was moved by Christian Medical College (CMC) in Vellore.
“The Phase 4 trials for mixing doses of Covaxin and Covishield will soon take place soon on healthy 300 volunteers,” they stated.
They explained that the purpose behind the study is to see whether a person can be given two different vaccine shots of Covishield and Covaxin to complete the inoculation course.
A section of experts advocated that using vaccines based on two different platforms could trigger a better immune response in a beneficiary.
The mix-and-match strategy is known as a heterologous prime and boost and has been used in the past against diseases such as Ebola and AIDS.
Traditionally, doses of the same vaccine are given multiple times as homologous boosts. New findings indicate that prime-boost can be done with different vaccines containing the same antigens. In many cases, such heterologous prime-boost can be more immunogenic than homologous prime-boost.
German Chancellor Angela Merkel had received a Moderna coronavirus vaccine as her second jab, after getting the first dose of AstraZeneca vaccine, bringing the spotlight once again on the mix-and-match of Covid-19 shots.



